Applications
Sorption Science is employed for a variety of applications across a range of industries, and our instruments are designed to reflect that.
As top-of-the-line sorption analyzers, our products can be employed across a huge range of applications, provide detailed and accurate data whatever your research purpose.

Our Applications

Carbon Capture Utilization and Storage
As one of the fastest-growing fields of research in new materials developments, being equipped to precisely and quickly evaluate a range of materials for their efficacy in Carbon Capture is essential to success.

Competitive Adsorption
Understanding the competitive sorption of water and organics is becoming increasingly important due to the implications of pollutants for overall health, and in industrial manufacturing processes. DVS is a sensitive technique which can measure two-component adsorption and competition, both in vapour phase and gas phase depending on the system being used.
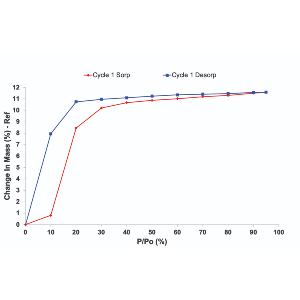
Water Sorption Isotherms and Kinetics
The water sorption kinetics of compounds and materials are integral to their physiochemistry and stability. Measuring a water sorption isotherm with DVS provides many insights into a material’s behavior, through classification of isotherms, examination of hysteresis, or the characterization of reversible or irreversible sorption processes.

Relative Humidity Induced Glass Transition Temperature
The effect of relative humidity on the glass transition temperature for amorphous compounds has a profound influence on the material’s physicochemical properties. Water acts as a plasticizer for many amorphous compounds, and the property changes initiated by a phase change can have a critical impact on the stability, performance, and shelf life of a product.

Formulation Performance
The formulation performance of a product can be affected in a variety of ways by moisture present in the air. The stability of these formulations is paramount to their efficacy. Both DVS and iGC-SEA can be used to examine how a product dries, deposition of product, adhesive properties of a powder blend, and the stabilization effects of protein stabilizers.
Vapor Pressure of Solids and Liquids
Vapor pressure is an important material physical property that defines the amount of vapor phase material that exists in equilibrium with the original material. All materials enter the vapor phase by sublimation (solid – gas) or evaporation (liquid – gas).

Surface Energy and Adhesion
Determination of the total surface energy of a solid can give useful indication of its overall activity. Work of cohesion and adhesion is a property proportional to surface energy. Evaluating cohesive forces at play within a powder sample can give a measure of how well powders can flow and their likelihood to aggregate.
Porosity and Surface Area
Characterizing the total surface energy profile of a solid is a useful tool for comparing the performance of similar character materials. It also provides a good starting point for estimating numerous other physicochemical properties such as aggregation likelihood, flowability etc.

Polymers – Solvent Swelling and Interactions
The world’s most advanced dual balance gravimetric vapor sorption instrument, the DVS Discovery combines an innovative dual balance design with outstanding vapor generation and temperature control. This increases high throughput while reducing operator time.

Drying and Dehydration
Matter under ambient conditions interacts with moisture and volatiles continuously. The exposure of heat and a high air flow over a material is likely to induce some form of drying. A material’s ability to resist rapid transitions between wet/hydrated states can be a good measure of stability.

Diffusion and Permeability
Diffusion and mass transfer play a significant role in a material’s performance and stability. Gases and vapors interact and exchange with a surface altering its external interfaces and internal bulk composition. The ability to closely monitor diffusion processes is essential in order to determine important characteristics like product lifetime and exchange efficiency.

Sorption Capacity
The sorption capacity of a material is related to a materials surface chemistry, surface area, and, if applicable, its pore size distribution. The sorption properties of a material can inform on all these properties, as well as develop a key understanding into a materials performance, lifespan, and activity.

Batch-to-Batch Variability
Whether due to the processing procedure, the amorphous content of the sample, or even if polymorphic varieties of a particular compound exist, batch-to-batch variation of active materials pose a significant problem across all industries. While chemically identical, variations may cause physicochemical instability and phase changes which need to be avoided at all stages of manufacturing.
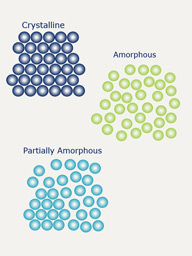
Amorphous Content Determination
Amorphous materials present complex and challenging difficulties associated with their processing, storage, and ultimate performance of the product. They have unique properties to their crystalline counterparts, and while amorphous content may be desired, it can also be generated during the processing of different solids via milling, spray drying or wet granulation.

Solubility
Solubility is a critical consideration across a wide range of industries. In pharmaceuticals, solubility is an incredibly important parameter needed to achieve the correct drug dosage in the body. Poor solubility is often associated with higher doses needed to reach the correct bioavailability of the drug. Solubility is affected by temperature, the solvent, and the properties of the compound undergoing dissolution.