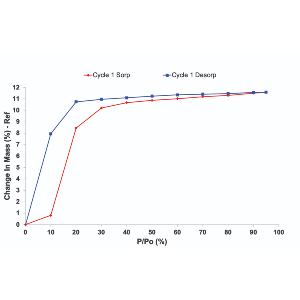
Solubility
Solubility is a critical consideration across a wide range of industries. In pharmaceuticals, solubility is an incredibly important parameter needed to achieve the correct drug dosage in the body. Poor solubility is often associated with higher doses needed to reach the correct bioavailability of the drug. Solubility is affected by temperature, the solvent, and the properties of the compound undergoing dissolution.


Understanding the solubility of a compound is needed early on in drug discovery and has wide-ranging implications throughout the whole drug discovery process. In other industries, the solubility of components in a formulation has important implications on the stability and shelf life of a product, such as in cosmetic or food formulation. It also has great importance in crystallization processes.

Measuring solubility with DVS & iGC
Our range of DVS & iGC products are perfectly suited to accurately measure solubility. View the range using the link below to find out more about which instrument is right for you.