Batch-to-Batch Variability
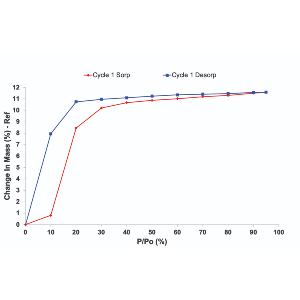
Whether due to the processing procedure, the amorphous content of the sample, or even if polymorphic varieties of a particular compound exist, batch-to-batch variation of active materials poses a significant problem across all industries. While chemically identical variations may cause physicochemical instability and phase changes, it needs to be avoided at all stages of manufacturing.
To understand the causes of these variations, it is important to first have sensitive techniques which can detect these differences between batches. Using our novel techniques and innovative instrumentations these variations can be detected at the most detailed level.


Solutions for batch-to-batch variation
Our DVS & iGC instrument provide unparalleled abilities for measuring and understanding batch-to-batch variation. If this application is an important part of your research, use the button below to view which of our instruments is right for you.